Figure 8 | Highly porous carboxylated activated carbon from jute stick for removal of Pb2+ from aqueous solution | SpringerLink

a) Optical spectra of calix-3NO2 (10 mg L -1 ) without and with the... | Download Scientific Diagram

SOLVED: EXAMPLE #4: DILUTIONS You want to make 200 ug/mL of ampicillin (an antibiotic) You have a 10 mglmL stock solution of ampicillin. How much would you need to add to make
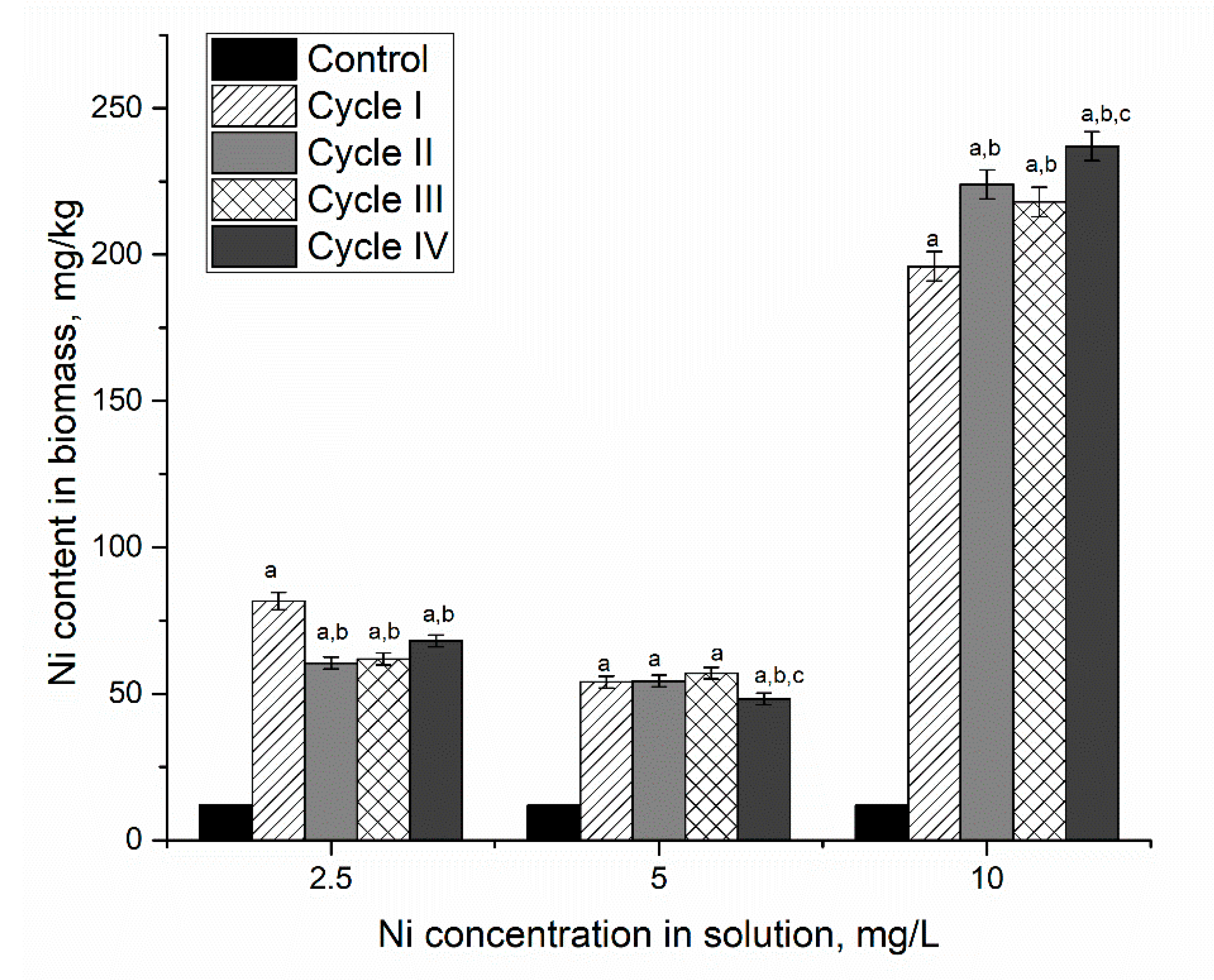
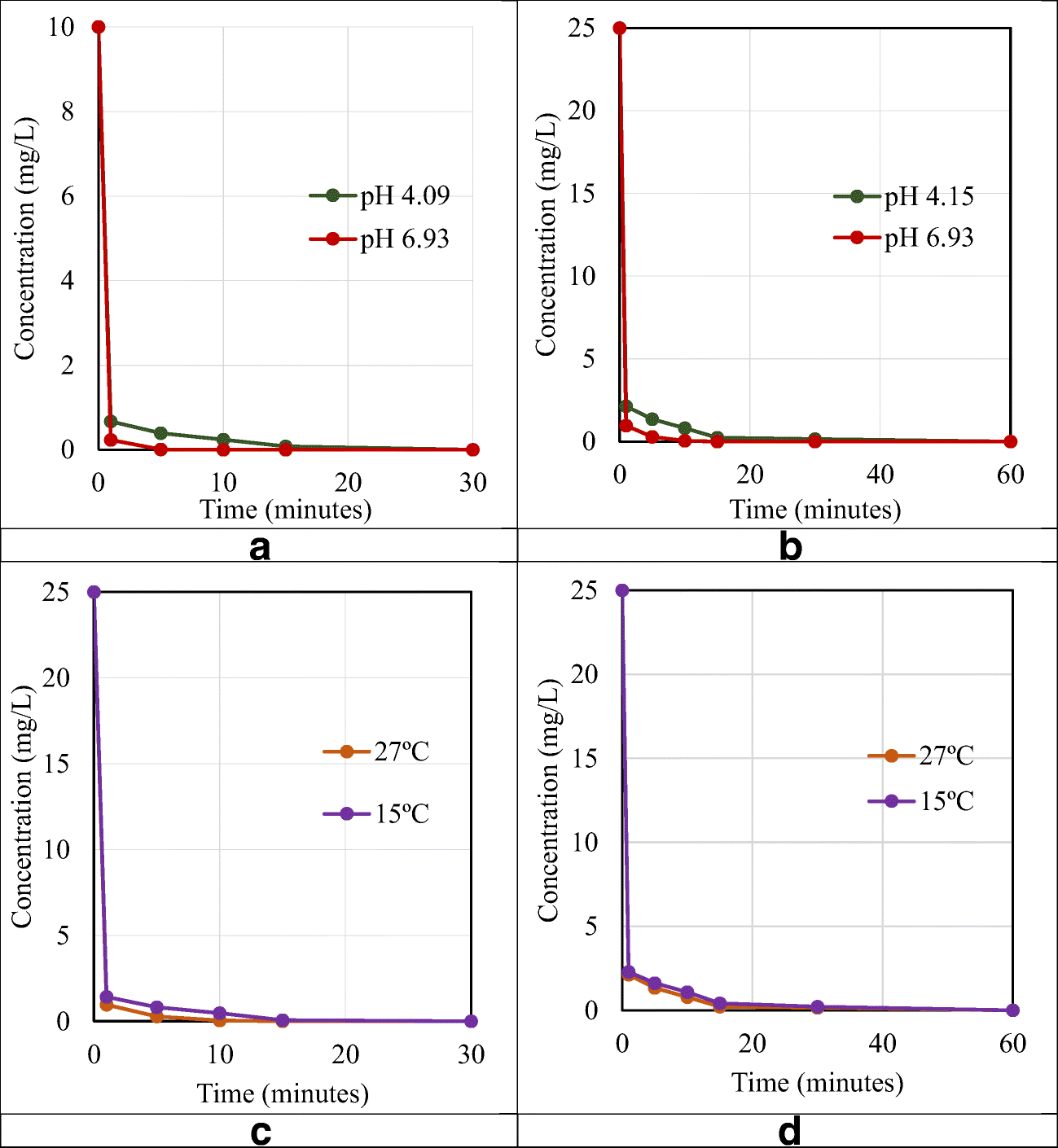
Microorganisms | Free Full-Text | Assessment of Metal Accumulation by Arthrospira platensis and Its Adaptation to Iterative Action of Nickel Mono- and Polymetallic Synthetic Effluents | HTML

OneClass: You have a 10 mg/ml stock solution of ethidium bromode and want a final concentration of 1 ...

Selective Reduction of Au(I) from a High-Concentration Thiosulfate Solution for Gold Recovery Using One-Step Heat-Treated High-Sulfur Coal | ACS Sustainable Chemistry & Engineering

Amazon.com: Hach 2374820 Lead Standard Solution, 10 mg/L as Pb (NIST), 25mL : Industrial & Scientific

SOLVED: A water sample contains 20 mg NO3-/L. What is the concentration in ( a) ppmm, (b) moles/L? ρH2O at 25 degrees Celsius = 997.8 g/L
40—THE EFFECTS OF SODIUM CHLORIDE, PYRIDINE AND ETHYL ALCOHOL ON THE ABSORPTION SPECTRA OF CERTAIN AZO DYES IN AQUEOUS SOLUTIO

M70800-10.0 - Murashige & Skoog Medium with MES Buffer and Vitamins, 49 Grams of Powder, Makes 10 Liters of Solution











