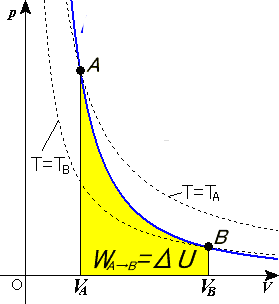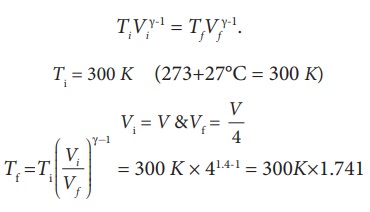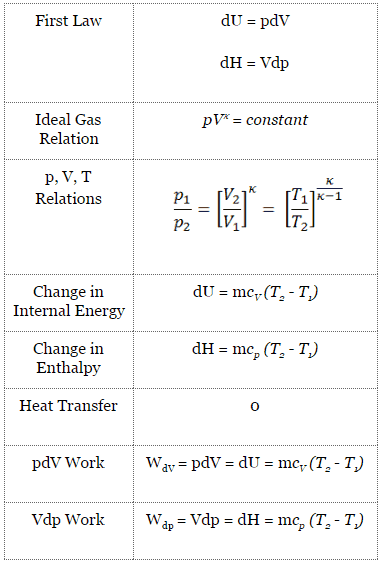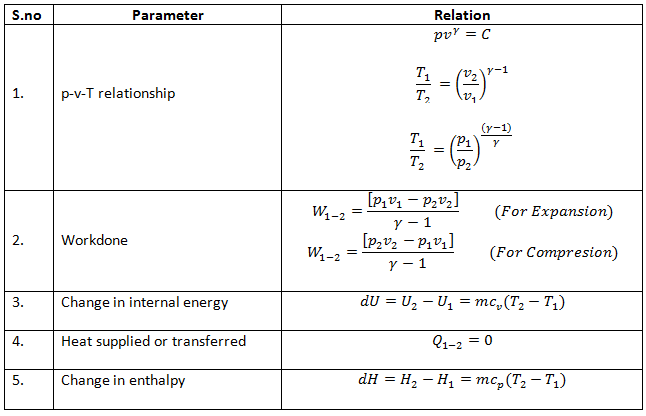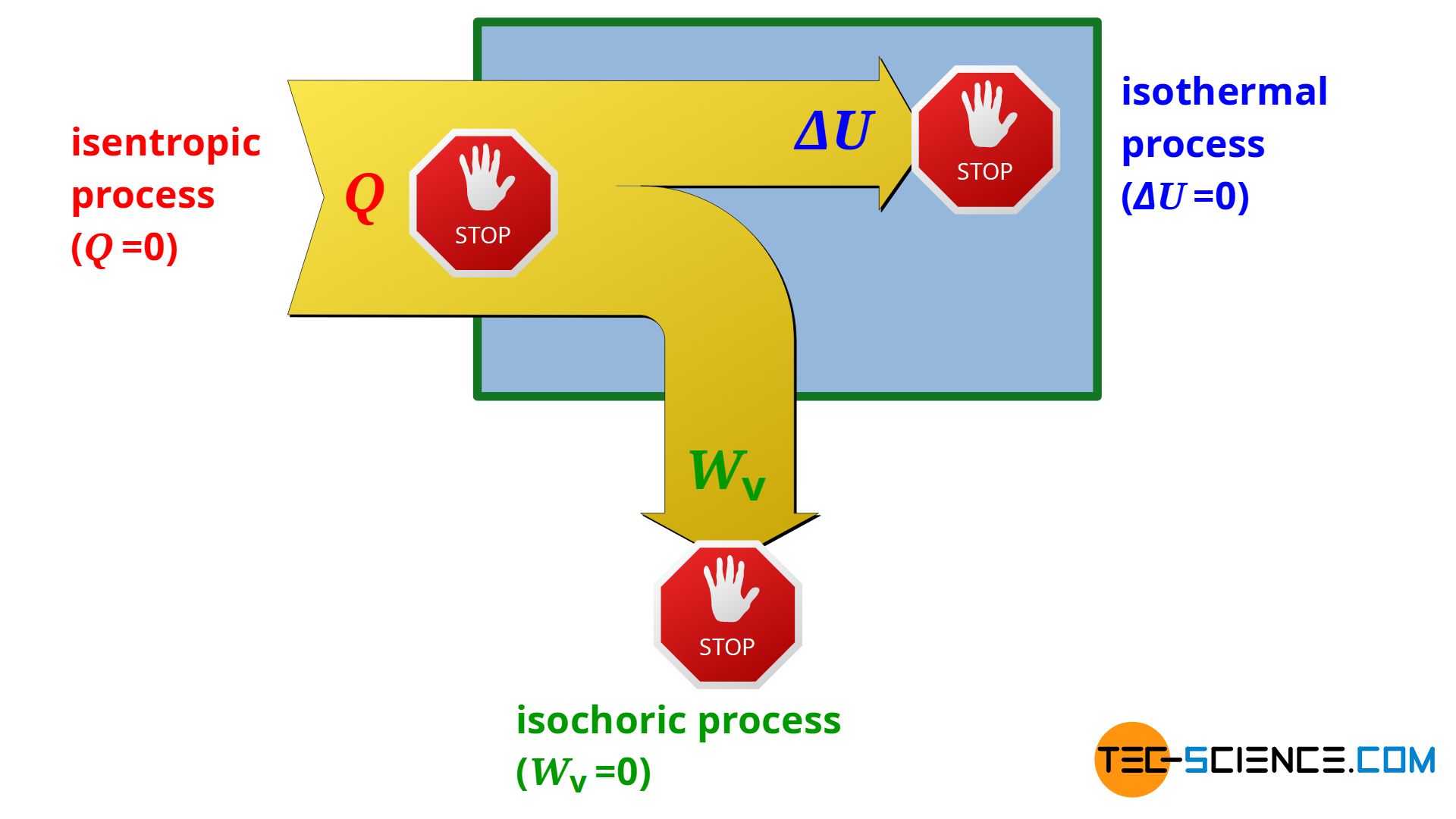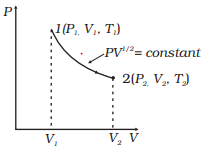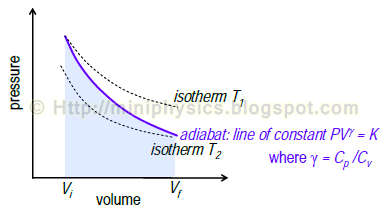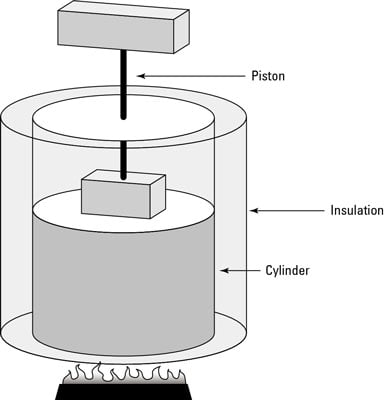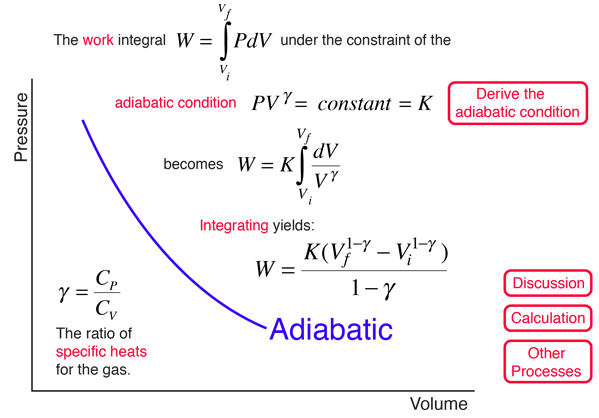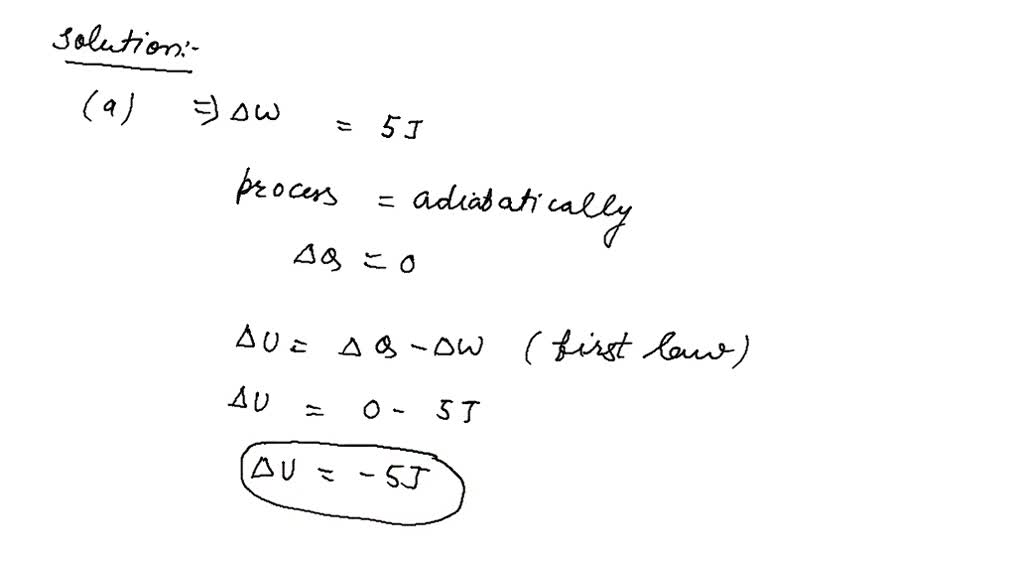
SOLVED:For each of the following adiabatic processes, find the change in internal energy. ( a ) A gas does 5 J of work while expanding adiabatically. (b) During an adiabatic compression, 80

homework and exercises - How do you calculate the change in temperature of an adiabatic system? - Physics Stack Exchange

Work Done in Adiabatic Process Calculator | Online Work Done in Adiabatic Process Calculator App/Software Converter – CalcTown